Shopping Cart
- Remove All
 Your shopping cart is currently empty
Your shopping cart is currently empty
Cathepsin B Protein, Human, Recombinant (His) is expressed in HEK293 mammalian cells with His tag. The predicted molecular weight is 37.2 kDa and the accession number is A0A024R374.

| Pack Size | Price | Availability | Quantity |
|---|---|---|---|
| 5 μg | $118 | In Stock | |
| 10 μg | $198 | In Stock | |
| 20 μg | $339 | 7-10 days | |
| 50 μg | $645 | In Stock | |
| 100 μg | Inquiry | 7-10 days |
| Biological Activity | Measured by its ability to cleave the fluorogenic peptide substrate Z-LR-AMC. The specific activity is >2,500 pmoles/min/μg. |
| Description | Cathepsin B Protein, Human, Recombinant (His) is expressed in HEK293 mammalian cells with His tag. The predicted molecular weight is 37.2 kDa and the accession number is A0A024R374. |
| Species | Human |
| Expression System | HEK293 Cells |
| Tag | C-His |
| Accession Number | P07858 |
| Synonyms | CPSB,cathepsin B,APPS |
| Construction | A DNA sequence encoding the pre pro form of human CTSB (Arg18-Ile339) (NP_001899.1) was expressed with a C-terminal polyhistidine tag. Predicted N terminal: Arg 18 & Phe 74 |
| Protein Purity | > 97 % as determined by SDS-PAGE 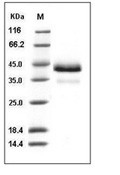 |
| Molecular Weight | 37.2 kDa (predicted); 36 and 43 kDa (reducing conditions) |
| Endotoxin | < 1.0 EU/μg of the protein as determined by the LAL method. |
| Formulation | Lyophilized from a solution filtered through a 0.22 μm filter, containing PBS, pH 7.4. Typically, a mixture containing 5% to 8% trehalose, mannitol, and 0.01% Tween 80 is incorporated as a protective agent before lyophilization. |
| Reconstitution | A Certificate of Analysis (CoA) containing reconstitution instructions is included with the products. Please refer to the CoA for detailed information. |
| Stability & Storage | It is recommended to store recombinant proteins at -20°C to -80°C for future use. Lyophilized powders can be stably stored for over 12 months, while liquid products can be stored for 6-12 months at -80°C. For reconstituted protein solutions, the solution can be stored at -20°C to -80°C for at least 3 months. Please avoid multiple freeze-thaw cycles and store products in aliquots. |
| Shipping | In general, Lyophilized powders are shipping with blue ice. |
| Research Background | Cathepsin B is a papain-family cysteine protease that is normally located in lysosomes, where it is involved in the turnover of proteins and plays various roles in maintaining the normal metabolism of cells. This protease has been implicated in pathological conditions, e.g., tumor progression and arthritis. In disease conditions, increases in the expression of cathepsin B occur at both the gene and protein levels. Cathepsin B is synthesized as a preproenzyme and the primary pathways for its normal trafficking to the lysosome utilize mannose 6-phosphate receptors (MPRs). Mature cathepsin B has the ability to degrade several extracellular matrix components at both neutral and acidic pH and has been implicated in the progression of several human and rodent tumors progression and arthritis. Cathepsin B expression is increased in many human cancers at the mRNA, protein and activity levels. It is also frequently overexpressed in premalignant lesions, an observation that associates this protease with local invasive stages of cancer. Increased expression of cathepsin B in primary cancers, and especially in preneoplastic lesions, suggests that this enzyme might have pro-apoptotic features. Active cathepsin B is also secreted from tumours, a mechanism likely to be facilitated by lysosomal exocytosis or extracellular processing by surface activators. Cathepsin B is localized to caveolae on the tumour surface, where binding to the annexin II heterotetramer occurs. Thus CTSB is suggested as a tumor marker. Additionally, Cathepsin B can degrade extracellular matrix proteins, such as collagen IV and laminin, and can activate the precursor form of urokinase plasminogen activator (uPA), perhaps thereby initiating an extracellular proteolytic cascade. |

Copyright © 2015-2025 TargetMol Chemicals Inc. All Rights Reserved.