Shopping Cart
- Remove All
 Your shopping cart is currently empty
Your shopping cart is currently empty
BCL-XL Protein, Human, Recombinant (His) is expressed in E. coli expression system with His tag. The predicted molecular weight is 25.2 kDa and the accession number is Q07817.

| Pack Size | Price | Availability | Quantity |
|---|---|---|---|
| 5 μg | $48 | 7-10 days | |
| 10 μg | $75 | 7-10 days | |
| 20 μg | $118 | 7-10 days | |
| 50 μg | $229 | 7-10 days | |
| 100 μg | $386 | In Stock |
| Biological Activity | 1. Measured by its binding ability in a functional ELISA.
2. Immobilized human BID at 10 μg/mL (100 μl/well) can bind biotinylated human BCL2L1, The EC50 of biotinylated human BCL2L1 is 7.1 ng/mL.
3. Immobilized mouse BID at 10 μg/mL (100 μl/well) can bind biotinylated human BCL2L1, The EC50 of biotinylated human BCL2L1 is 7.01 ng/mL. |
| Description | BCL-XL Protein, Human, Recombinant (His) is expressed in E. coli expression system with His tag. The predicted molecular weight is 25.2 kDa and the accession number is Q07817. |
| Species | Human |
| Expression System | E. coli |
| Tag | C-His |
| Accession Number | Q07817 |
| Synonyms | PPP1R52,bcl-xS,BCLXS,BCL-XL/S,bcl-xL,BCLXL,Bcl-X,BCLX,BCL2-like 1,BCL2L |
| Construction | A DNA sequence encoding the human BCL2L1 (NP_612815.1) (Met1-Arg212) was fused with a polyhistidine tag at the C-terminus. Predicted N terminal: Met 1 |
| Protein Purity | > 85% as determined by SDS-PAGE 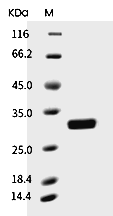 |
| Molecular Weight | 25.2 kDa (predicted); 30.5 kDa (reducing conditions) |
| Endotoxin | Please contact us for more information. |
| Formulation | Lyophilized from a solution filtered through a 0.22 μm filter, containing 5 mM Tris, 1% Glycerol, pH 8. Typically, a mixture containing 5% to 8% trehalose, mannitol, and 0.01% Tween 80 is incorporated as a protective agent before lyophilization. |
| Reconstitution | A Certificate of Analysis (CoA) containing reconstitution instructions is included with the products. Please refer to the CoA for detailed information. |
| Stability & Storage | It is recommended to store recombinant proteins at -20°C to -80°C for future use. Lyophilized powders can be stably stored for over 12 months, while liquid products can be stored for 6-12 months at -80°C. For reconstituted protein solutions, the solution can be stored at -20°C to -80°C for at least 3 months. Please avoid multiple freeze-thaw cycles and store products in aliquots. |
| Shipping | In general, Lyophilized powders are shipping with blue ice. |
| Research Background | B-cell lymphoma-extra large (Bcl-xl) is a transmembrane molecule in the mitochondria. Bcl-xL (BCL2L1), belongs to the Bcl-2 family. Members of the bcl-2 family encode proteins that function either to promote or to inhibit apoptosis. Antiapoptotic members such as Bcl-2 and Bcl-xL prevent PCD in response to a wide variety of stimuli to take part in cancer survival. Conversely, proapoptotic proteins, exemplified by Bax and Bak, can accelerate death and in some instances are sufficient to cause apoptosis independent of additional signals. The crystal and solution structures of a Bcl-2 family member, Bcl-xL is like this: The structures consist of two central, primarily hydrophobic α-helices, which are surrounded by amphipathic helices. A 60-residue loop connecting helices αl and α2 was found to be flexible and non-essential for anti-apoptotic activity. Bcl-xL is characterized as an important factor in autophagy, inhibiting Beclin 1-mediated autophagy by binding to Beclin 1. In addition, Beclin 1, Bcl-2 and Bcl-xL can cooperate with Atg5 or Ca2+to regulate both autophagy and apoptosis. Bcl-xL is also implicated in anoxia induced cell death. The pathway is initiated by the loss of function of the prosurvival Bcl-2 family members Mcl-1 and Bcl-2 / Bcl-XL, resulting in Bax- or Bak-dependent release of cytochrome c and subsequent caspase-9-dependent cell death. Thus, Bcl-xL, the well-characterized apoptosis guards, appears to be important in cell death. |

Copyright © 2015-2025 TargetMol Chemicals Inc. All Rights Reserved.